by Roelof D Schuiling and Poppe L de Boer
Background
Almost all of the CO2 that has ever leaked out of the planet has been removed from the atmosphere and the ocean, and sustainably stored in rocks, mainly by weathering, and also in the later part of the Earth’s history by storage as organic carbon. During weathering, which is the reaction of rocks with CO2 and water, CO2 is first converted to bicarbonate solutions. In the ocean corals, shellfish, and plankton convert them to carbonate sediments, which form the ultimate sustainable storage of CO2 (Figure 1).
 |
| Figure 1. A Karst landscape in China is one of the many stores for CO2 of the world. |
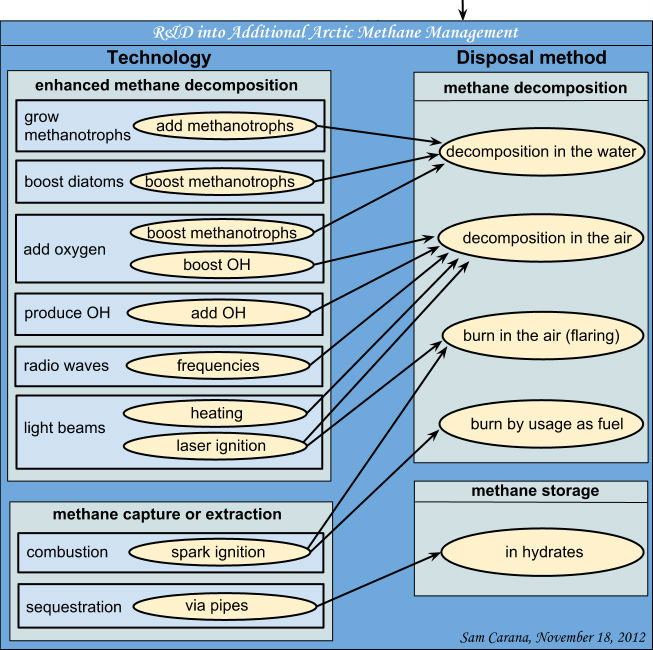
Six solutions
Six low-cost or financially self-supporting ways are described in which we can store large
volumes of CO2 and/or significantly diminish CO2 emissions:
- Nickel Farming: A switch from nickel mining, ore dressing, and nickel recovery to nickel farming by the use of nickel hyperaccumulator plants. This switch will cut down CO2 emissions because it avoids the energy-intensive steps in the nickel cycle and enhances the weathering of olivine or serpentine which captures additional CO2.
- Biodiesel from Diatoms: Diatoms contain approximately 50% of lipids, which makes them an ideal starting material for the production of biodiesel. They grow fast, provided they have a source of silica. They do not suffer from drawbacks of land-grown biofuel crops. They do not occupy vast tracts of land that are urgently needed for food production, and they do not require vast amounts of scarce irrigation water and fertilizer. The use of biodiesel from diatoms will reduce the CO2 emissions from fossil fuels.
- Quenching Forest Fires: Forest fires are the second largest emitter of CO2, after fossil fuels. It was demonstrated that quenching such fires with a slurry of serpentine powder is considerably more effective than quenching with water. This reduces the emissions of CO2 by the fires and the associated financial losses. The serpentine that was calcined by the fire reacts very fast with CO2 and water afterwards, thereby compensating part of the emitted CO2 during the fire. A better and quicker mastery over forest fires may also help to save lives.
- Supergreen Energy: If the heat that is released by the weathering of olivine is trapped, this would represent a huge alternative source of energy that additionally captures large volumes of CO2, hence the name supergreen energy. A basic scenario is described how this could be achieved.
- Coastal Protection: When olivine is used for coastal protection (breakwaters, artificial reefs, sand replenishment on beaches) this has a direct effect against ocean acidification. CO2 is absorbed as bicarbonate, and the pH of the surrounding waters rises.
- Olivine in High-Energy Marine Environments: Large areas of shallow seas are subjected to strong currents that can transport gravel. When olivine grit is spread on the sea floor, the grains are kept in motion and bump and rub against each other. This destroys reaction-limiting silica coatings on the grain surfaces and releases micronsized slivers that rapidly react with sea water. It is the most direct way to counter ocean acidification.
1. Nickel Farming
All mining operations have an impact on the environment. This also holds for nickel, independent of the type of ore, whether nickel laterite or nickel sulfide. Nickel laterites must be leached and nickel sulfides must be roasted and dissolved. These steps are energyintensive and polluting. These disadvantages can be reduced if part of the world nickel production is gradually replaced by a switch to nickel farming. A fairly large number of plant species from different families are known to exhibit the remarkable property that they very effectively extract nickel from nickel-rich soils and store it in their tissues (Figure 2). Soils on serpentinized peridotites often contain no more than 0.2% of nickel, but the ash of these plants may contain 10% or more of nickel, much richer than the richest nickel ores. If some NPK fertilizer is spread over nickel-rich soils and such plants are sown, these nickel hyperaccumulator plants can be harvested at the end of the growing season, and their nickel content can be recovered after ashing. Several of these plants are perennial, so they do not need to be sown every year.
 |
| Figure 2. Alyssum corsicum, a nickel hyperaccumulator. |
A first estimate [1] shows that the recovery of nickel by using these plants will cost no more than the current way of nickel production. This means that all the savings on CO2 expenditure and CO2 storage are essentially cost-free. In instances where an appropriate value may be associated with the CO2 savings compared to conventional nickel production, nickel farming may economically outcompete incumbent nickel production processes.
Once such nickel hyperaccumulation systems will have been fully developed and become deployable, it is hoped that governments adopt incentive structures that oblige mining companies with nickel mining assets to conduct at least part of their businesses with these methods and that associated CO2 savings and removals are quantified and verified. In addition, because the nickel obtained from phytomining is not extracted from nickel ores but from common peridotite rocks, nickel farming will extend the lifetime of nickel ore deposits.
2. Biodiesel from Diatoms
Diatoms (siliceous algae) make up a large part of the biomass in the oceans. They consist about 50% of the lipids, which makes them an ideal raw material for the production of biodiesel. The process to make biodiesel from algae is already known. They grow fast and can outcompete their competitors in the algal world in the fight for food, provided that there is sufficient silica available in their environment. Diatoms need silica for the construction of their silica skeleton (Figure 3). An extensive discussion of the role of dissolved silica in promoting the growth of diatoms at the cost of other plankton like dinoflagellates can be found in [2].
 |
| Figure 3. Diatoms have delicate silica skeletons. |
Diatoms do not have these drawbacks, but before they can be used as an alternative source of biofuel, the problems of mass culturing and harvesting them must be solved. A large-scale mariculture of diatoms might take the following shape. An artificial lagoon can be sectioned off by a dike around a sector of shallow seawater in front of a beach section.
The beach between the low-tide mark and the high-tide mark must be covered by a layer of olivine sand with a thickness of about 0.5 m. One or more U-shaped tubes are left in the dike that connects the lagoon with the open sea, permitting the tide to reach the lagoon and to alternatively wet and drain the olivine beach. These tubes should be closed by a perforated metal plate covered with a plankton net. This would permit the exchange of water, but prevent the diatoms to be carried out of the lagoon by the ebb. The olivine will weather, and the weathering solution, including the silica that is set free during the olivine reaction, will be distributed in the lagoon. In addition, the bicarbonate that is captured during olivine weathering will be used by the diatoms for photosynthesis.
When the silica limitation is removed, diatoms will form a quasi-monoculture in the lagoon. Nutrients should, of course, be added, mainly for their ammonia and phosphate requirements. A cheap way to do this would be by the use of struvite, an ammonium-magnesium phosphate that is produced by a simple and robust technology in the treatment of organic wastes, including manure, urban waste, and urine [3]. Struvite is a slow-release fertilizer that will steadily add ammonium and phosphate to the lagoon. The addition of nutrients should be limited, however, because diatoms react to a slight starvation by raising their lipid content, which increases their value for biodiesel production. The diatom production can be increased by underwater lighting at night.
Harvesting the diatoms efficiently is a major problem. The following possibility may provide a solution. Dig a hole inside the lagoon. Dead diatoms will collect in this pit, also thanks to the fact that they are relatively heavy due to their silica skeletons. From time to time, this mass of dead diatoms can be sucked up, drained and transported to the biofuel plant.
When the culture and harvesting of vast volumes of diatoms can be successfully accomplished, this application will become financially self-supporting and will reduce CO2 emissions from the burning of fossil fuels. It can be setup in any country with marine coastlines, preferentially in dry climate zones with abundant sunshine.
3. Quenching Forest Fires
Forest fires (Figure 4) are the largest CO2 emitters after the burning of fossil fuels. Forest fires and, to a lesser extent, other forest losses account annually for about 6 Gt of extra CO2 emissions on a total of somewhat more than 30 Gt of human CO2 emission [4]. They cause every year not only huge financial losses but also the deplorable loss of human lives. Experimental fires at the test site of Brandbeveiliging BV (Fire Protection) in the Netherlands were considerably faster and completely extinguished by spraying with a suspension of serpentine powder than with plain water. Serpentinite powder from the PASEK mine in North-West Spain and from the Isomag Mine in Austria was used with equally positive results.
 |
| Figure 4. Forest fires are the second largest emitter of CO2 in the world. |
So, when forest fires are raging, the spraying of serpentine slurries (almost as simple as spraying water, because a 40% serpentine slurry is still very fluid) can reduce the extent and severity of such fires. When a reduction of 10% in forest losses could be achieved worldwide, this would already be a major breakthrough, since this represents a reduction of 0.6 Gt of CO2 emissions each year.
Moreover, after extinction of the fire, the calcined serpentine will quickly react with CO2 and the first rainwater, thereby compensating part of the CO2 that was emitted by the fire. It is clear that the spraying of serpentine (serpentine powder is a cheap and ubiquitous material) is a very cost-effective way of reducing the huge financial losses from forest fires, and it holds the promise of reducing losses of life as well. It pays amply for the reduction in CO2 emission by limiting the areal extent of burnt forest and by the capture of CO2 by the reaction of the calcined serpentine afterwards. It also limits the required volumes of water considerably, which is important in hot dry summers in countries that are most vulnerable for forest fires and have only limited fresh water resources.
It should be considered whether the spraying of serpentine slurries can also be used in the containment of tunnel fires.
4. Supergreen Energy
A property of olivine weathering that is commonly overlooked is its energy production. When olivine is weathering under conditions of limited water flow, it weathers according to:
Mg2SiO4 + CO2 + H2O → Mg3Si2O5(OH)4 + MgCO3
Olivine, Carbon dioxide, Water Serpentine, Magnesite
In a system that is very well isolated and has a large volume-to-surface ratio, it might be possible to recover most of that energy. Rocks are excellent thermal isolators, as shown by caves. If one visits a cave in summer, it feels nice and cool, and in winter it feels pleasantly warm. This is because the surrounding rocks provide a good thermal isolation and keep the cave at a fairly constant temperature throughout the year. The larger the volume of olivine sand under good isolating conditions, the better it will be able to develop and keep a high temperature. One might say, volume stands for heat production and surface area stands for heat loss; thus, the larger the volume (and the thicker the isolation), the lesser the heat loss.
A scenario that provides these conditions could be the following. An existing 550-m deep lignite mine in Germany (Figure 5) will be taken as an example; but in fact, any deep open pit mine could serve, whether in operation or left as a scar in the landscape after closure .
 |
| Figure 5. A lignite mine in Germany. |
As long as the temperature is low, the reaction will be slow. In order to kickstart the process, it is advisable to first inject steam to heat the inside of the mass. This will increase the reaction rate, and as the reaction takes off, the temperature will rise further and the reaction accelerates.
When the system has reached a sufficiently high temperature to be of interest for power production, water is passed through the heat exchangers and converted to high-pressure steam.
It should be evident that such a system will require a lot of additional and rather unusual engineering before it can be operational. On the other hand, the potential reward is huge because it represents an almost unlimited amount of energy. This energy is called supergreen energy because it does not produce CO2, but, on the contrary, it traps it in a safe and solid form. The question asked by the author in [6] is relevant ‘So what would we prefer, a CCS infrastructure that uses a quarter of a power station’s electricity to sequester its CO2 emissions under the North Sea or one that generates additional electricity and useful materials products?’.
A major technical problem may arise if silica that is released during the olivine reaction would form a layer on the olivine grains, preventing the reaction to proceed. A possible way out is to mix the olivine sand with some minute quartz grains. Quartz has a much lower solubility than amorphous silica, so the dissolved silica that is released in solution will tend to diffuse to the quartz grains and precipitate as an overgrowth on quartz surfaces instead of on the olivine grains, leaving the olivine surfaces free for continued reaction.
5. Coastal Protection
Olivine can be used in several ways to protect coastlines against erosion. Olivine is considerably heavier than normal quartz sand (specific masses of 3.4 versus 2.65 kg/m3), which makes it more resistant to physical erosion. Olivine blocks can be used in the construction of permeable breakwaters. In a permeable triangular breakwater, pointing into the sea, the force of the longshore (flood and ebb) currents is weakened because part of the water passes through the breakwater and loses momentum in doing so, while another part is deviated from the coast. Both effects reduce coastal erosion. If the sections at either side of the breakwater are covered with olivine sand, it will resist erosion even better.
Another way of using olivine for coastal protection is the construction of olivine reefs at strategic points to keep waves and currents away from the coast. If the seawater that is enclosed in the reefs is only slowly refreshed, its pH will rise as a consequence of the olivine reaction. This may lead to the precipitation of calcite, so that these reefs are self-cementing. They will become hatching and hiding places for fish and a place for mussels and oysters to settle (Figure 6).
 |
| Figure 6. The sea as a threat: the Hondsbossche Zeewering along the Dutch Coast. |
 |
Figure 7. The sea as an ally. Children making sand sculptures of olivine sand that will merge with the sea at high tide and help in counteracting ocean acidification. |
 |
| Figure 8. Sixty-meter tall aragonite (replacing brucite) chimneys on Lost City seamount. |
6. Olivine in High-Energy Marine Environments
It is a paradigm that weathering on land, and under marine conditions, always would be a slow process. When olivine grains, preferably of different sizes, are free to be kept in motion by currents, their weathering is a fast process. The grains are quickly rounded and abraded by mutual collisions (Figure 9), producing myriads of micron-sized slivers (see picture in Additional file 1; see also [9]).
 |
| Figure 9. Angular olivine grains are quickly rounded and abraded by mutual collisions when kept in motion. |
Many shallow sea floors are covered with gravel. When 700,000 km2 of such sea bottoms are covered each year with a 1-cm thick layer of olivine grit, this would compensate the entire anthropogenic CO2 emissions, and raise the pH of the oceans. To make it more concrete, the following example may serve. Part of the continental shelf between the Shetland Isles and France, i.e. the Southern Bight of the North Sea, the English Channel and the Irish Sea, is covered with sand waves, and in and around the Channel, an area of well over 100,000 km2 experiences bed stresses capable of transporting gravel [10,11]. If a volume of 0.35 km3 coarse olivine is spread over 35,000 km2 of this area, this would compensate 5% of the world’s CO2 emissions, that is more than the combined emissions of the adjoining countries, the UK, France, Ireland, Belgium and the Netherlands together [9].
Another site where the spreading of coarse olivine grit may work out well is the Maelstrom, with very strong tidal current in the Lofoten Islands, Norway, and there are many more suitable areas in shallow shelf seas.
The alkalinity brought in by the olivine is of great importance. It counteracts ocean acidification, and the contained bio-limiting nutrients, Si and Fe, enhance marine productivity thereby capturing additional CO2. Another factor that makes this approach low-cost is that large carriers can bring the olivine directly to the place of use, where they are discharged, thus avoiding harbor dues and additional transport costs.
Results and discussion
A preliminary volume and cost-benefit estimate
At this early stage, it is virtually impossible to provide accurate estimates of the volumes of CO2 involved for each of the options, and of the amount of money potentially won or lost.
Table 1 should, therefore, be taken as a not too-educated guess of the orders of magnitude involved in each of the six options. The large spread in the numbers for the first five options is caused by the uncertainty whether the particular activity will be executed in a few tests on essentially pilot scale, or as a worldwide activity.
Table 1. Estimated order of magnitude of CO2 capture and/or emission reduction and money involved
| CO2 capture or emission reduction | Cost or benefit | |
| Unit | 1 Million ton | 1 Million euro |
| Options: | ||
| 1. Nickel farming | 1 to 50 | 0 to +200 |
| 2. Biodiesel from diatoms | 50 to 1,000 | +10 to +500 |
| 3. Quenching forest fires | 100 to 1,000 | +200 to + 2,000 |
| 4. Supergreen energy | 20 to 1,000 | +50 to + 5,000 |
| 5. Coastal protection | 10 to 1,000 | −1 to + 100 |
| 6. Olivine in high-energy waters | 25,000 | −500,0003 |
The cost of the olivine in high-energy shallow seas is calculated as the total costs of spreading 25 Gt of crushed olivine in shallow high-energy seas. When compared to carbon capture and storage (CCS), it should not be marked as a cost of 50 billion euro, but as a benefit of 0 billion euro.
Conclusions
It is likely that the first five examples of large-scale applications of the olivine option that are presented in this paper will all turn out to be profitable or, at least, financially self-supporting without requiring subsidies or carbon credits. The costs/benefits of the spreading of olivine in high-energy shallow seas depend on the way to calculate it. If it is just the cost of the operation itself, this total solution of the climate problem and ocean acidification costs a lot of money (order of 15% of the price of the equivalent amount of crude oil), but if it is compared to the costs of the CCS alternative, which is still on the agenda of several governments, it will save a huge amount of money. The major obstacle may well be that the unusual character of the proposals will delay their introduction because parties have a tendency to shy away from untested innovative approaches. Each of the six represents a major breakthrough in the attempts to control climate change and ocean acidification.
Methods
This study utilized stimulation of a chemical reaction that has been common at the Earth’s surface over the last 4.5 billion years.
Competing interests
The authors declare that they have no competing interests.
Authors’ contributions
RDS developed ideas about the use of stimulated olivine weathering as a means to counter human CO2 emissions. PDB carried out flume experiments. Both authors contributed to, read and approved the final manuscript.
Acknowledgements
Prof. Elburg (Durban) is thanked for suggesting some significant modifications. David Addison from Virgin Group, London is thanked for going through the text and suggesting a number of clearer formulations.
References
- Schuiling RD: Farming nickel from non-ore deposits, combined with CO2 sequestration. Natural Science 2013, 5:4.
- Scheffran J, Dürr HH, Wolf-Gladrow DA, De La Rocha CL, Köhler P, Renforth P, Joshua West A, Hartmann J: Enhanced chemical weathering as a geoengineering strategy to reduce atmospheric carbon dioxide, supply nutrients, and mitigate ocean acidification. Rev. Geophysics 2013, 51:113–149.
- Schuiling RD, Andrade A: Recovery of struvite from calf manure. Environ. Techn 1999, 20:765–768.
- Van der Werf GR, Morton DC, DeFries RC, Olivier JGJ, Kasibhatla PS, Jackson RB, Collatz GJ, Randerson JT: CO2 emissions from forest loss. Nature Geoscience November 2009, 2009:2.
- Schuiling RD: Serpentinization as a possible cause of high heat-flow values in and near oceanic ridges. Nature 1964, 201:807–808. no 4921.
- Priestnall M: Making money from mineralization of CO2. Carbon Capture Journal, February 03, 2013.
- Schuiling RD, de Boer PL: Rolling stones; fast weathering of olivine in shallow seas for cost-effective CO2 capture and mitigation of global warming and ocean acidification. Earth Syst. Dynam. Discuss 2011, 2:551–568. doi:10.5194/esdd-2-551-2011.
- Shipboard Scientific Party, Roe KR, Schrenk MO, Olson EJ, Lilley MD, Butterfield DA, Jeff G, Gretchen F-G, Blackman DK, Karson JA, Kelley DS: An off-axis hydrothermal vent field discovered near the Mid-Atlantic Ridge at 30°N. Nature 2001, 412:145–149.
- de Boer PL, Schuiling RD: Fast weathering of olivine in high-energy shallow seas for cost-effective CO2 capture as a cheap alternative for CCS, and effective mitigation of ocean acidification. AGU 2013 Fall Meeting, OS13A-1689. ftp://ftp.geog.uu.nl/pub/posters/2013/Mitigation_of_CO2_emissions_by_stimulated_natural_rock_weathering%e2%80%93fast_weathering_of_olivine_in_high-energy_shallow_seas-Schuiling_deBoer-November2013.pdf
- Belderson RH, Wilson RH, Holme NA: Direct observation of longitudinal furrows in gravel, and their transition with sand ribbons of strongly tidal seas. In Tide-Influenced Sedimentary Environments and Facies. Edited by de Boer PL, et al. Dordrecht: Reidel; 1988:79–90.
- Mitchell AJ, Ulicny D, Hampson GJ, Allison PA, Gorman GJ, Piggott MD, Wells MR, Pain CC: Modelling tidal current-induced bed shear stress and palaeocirculation in an epicontinental seaway: the Bohemian Cretaceous Basin, Central Europe. Sedimentology 2012, 57:359–388.
- McKinsey & Company: Carbon Capture & Storage: Assessing the Economics; Report September 22, 2008.
© 2013 Schuiling and de Boer
This article was published December 21, 2013, at enveurope.com/content/25/1/35 under a Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Post by Sam Carana.